Over the past decade, our industry has witnessed the scientific promise of cell and gene therapies. Patients with rare diseases or hard-to-treat diagnoses now have new treatment options harnessing human cells and genes to alter disease. But the accessibility of these therapies the industry has developed remains constrained not by what’s biologically possible, but how they are designed and manufactured.
The field has reached an inflection point. We’ve demonstrated the scientific foundation and its curative potential. But to make advanced therapies sustainable as a pillar of medicine, we must make them more accessible. The companies that will define cell and gene therapy’s future will be those who can eliminate the distance between top science and efficient manufacturing.
Integration of Manufacturing and Therapeutic Design
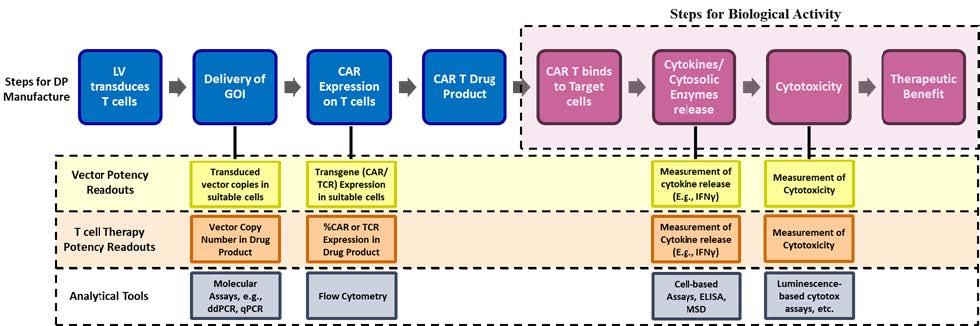
Traditional small molecule drug development has very siloed development pathways: a therapeutic is designed and developed by one team and then manufactured by another. This approach is challenging in cell and gene therapy, often leading to delays, setbacks, or even outright failures. We built ElevateBio to solve this problem with a new approach, one in which therapeutic design and manufacturing operate as an integrated ecosystem.
ElevateBio BaseCamp, our cGMP manufacturing business, goes beyond a traditional CDMO. We bring together expertise, advanced technologies, and state-of-the-art facilities to serve as a skilled partner to biopharmaceutical companies. This includes in-house manufacturing, process and analytical development, and quality control teams, all working in parallel to achieve tighter coordination and faster turnaround times. BaseCamp has industrialized genetic medicine manufacturing, achieving a 98% batch success rate across advanced programs.
Yet sustaining this success – and expanding it across new modalities – requires more than technical excellence alone.
Designing for Manufacturability from Day One
The future of cell and gene therapy depends on therapies designed with manufacturability in mind from the start – and into every stage of design. That’s why our team of process development scientists are embedded in design conversations early, creating commercial-ready processes in parallel with therapeutic development. Manufacturing insights flow back to inform molecular engineering in real time.
This includes integrating compact constructs and delivery systems engineered for both efficacy and efficiency. We apply scale-down and scale-up models to optimize performance, ensuring processes are fully scalable to GMP manufacturing and capable of meeting global demand.
We take the same approach through ElevateBio Life Edit, our gene editing technologies and R&D business. When our teams develop gene editors across all modalities, manufacturability is a design criterion from day one – not a constraint discovered in late-stage clinical trials. And by having BaseCamp and Life Edit sit alongside one another, we’re ensuring the latest manufacturing developments and insights flow back to inform R&D – and vice versa.
A Foundation for an Industry to Prosper
Looking beyond the science, what does a sustainable cell and gene therapy ecosystem require?
It’s more than better therapeutics. We need more treatment centers, expanding from dozens to hundreds for better patient access. The industry needs new commercial models that make advanced therapies economically viable for health systems. We need a whole new infrastructure where cell and gene therapy can become the standard of care for previously untreatable conditions.
But that infrastructure can’t be built upon unreliable manufacturing. We as an industry need to build a strong foundation – one built by designing, optimizing, and validating processes that reliably move therapies from bench to bedside. Without that foundation, the ecosystem simply can’t scale. And the window to build it is narrowing.
CAR-T is expanding into autoimmune indications with patient populations 10 times larger than oncology. In vivo therapies are advancing as new-generation modalities are adding layers of complexity. To support this growth, the field needs manufacturing designed for reliability and scale from day one.
Building What Comes Next
The field now needs the operational discipline and integrated thinking to deliver on that promise at population scale.
The therapies we’re developing today have the potential to transform millions of lives. But only if we build the systems to make them accessible, reliable, and sustainable.
At ElevateBio, we’re building that foundation by combining BaseCamp’s manufacturing platform with Life Edit’s R&D capabilities – and embedding therapeutic design expertise throughout. By doing so, we’ve created an integrated approach that’s building cell and gene therapy’s future and making a tangible impact for patients worldwide.