Gene Editing Design & Optimization
Translating gene editing concepts into optimized, clinic-ready therapeutic candidates.



// Gene Editing and
Engineering R&D Services
From Target to Clinical Lead
Our scientists have designed, discovered, and engineered versatile gene editing systems across five distinct editing modalities and built deep expertise in what it takes to move a program from early feasibility to a clinic-ready lead. Partners get access to that experience alongside one of the industry’s broadest editing portfolios, with programs structured to generate decision-enabling data at every stage.
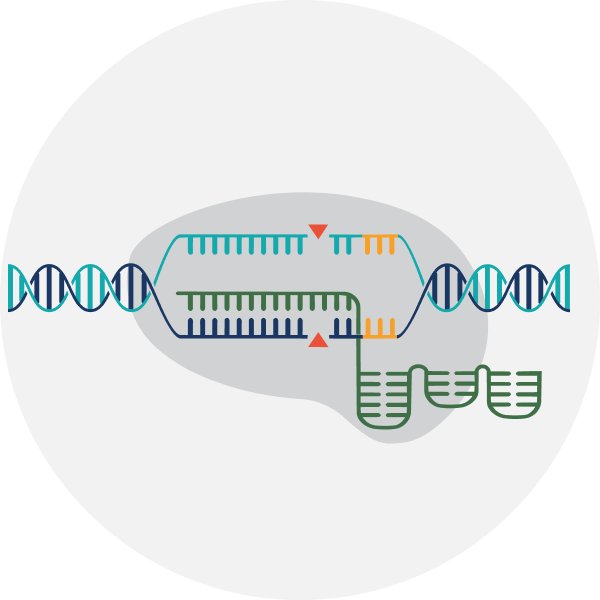
Five Modalities in a Single Platform
Our editing toolbox features CRISPR systems and editing technologies – including nuclease editing, base editing, reverse transcriptase editing, epigenetic editing, and targeted gene insertion – for both ex vivo and in vivo applications. That range of tools, coupled with delivery and engineering expertise, is what allows us to match the right editing approach to each target.
ElevateBio’s Gene Editing Design & Optimization Capabilities
Candidate DiscoveryEvery target presents a different editing challenge, and the right modality isn’t always obvious at the outset. We can design and evaluate multiple relevant editing strategies in parallel — nuclease editing, base editing, reverse transcriptase editing, epigenetic editing, and targeted gene insertion — and assess each against the biological requirements for the target, including gene locus accessibility, edit type, and downstream therapeutic goals. |  |
Candidate EngineeringWe optimize candidates for therapeutic application — guide RNA design, editor protein engineering, and construct refinement to improve specificity, potency, expression, and manufacturability. Our compact editors and diverse PAM recognition sequences give partners a broader design space than conventional systems, and engineering decisions are made with delivery, manufacturing, and regulatory expectations in mind from the start. |  |
Specificity AssessmentSpecificity and potency evaluation run alongside candidate engineering, so specificity data informs lead selection. We combine computational prediction with empirical methods to identify, quantify, and assess unintended edits across the genome, generating the datasets that support IND-enabling packages and regulatory interactions. |  |
Proof of ConceptFor partners evaluating whether a gene editing approach is viable for a specific target, our proof-of-concept work provides rapid, focused clarity — typically in 3 to 6 months. We run genomic analysis to identify viable editing strategies, design and evaluate editors across relevant modalities, and validate with an initial specificity assessment. Partners come out of this stage with active editors and supporting data to make a confident go/no-go decision before committing to further development. |  |
Clinical Lead OptimizationWith screening, engineering, and specificity data in hand, we refine the lead candidate — balancing potency, specificity, deliverability, and manufacturability — optimized through an IND-oriented process. This includes down-selection of editing approach, optimization of potency and specificity at the target locus, and safety and off-target characterization. Partners come out of this stage with a characterized lead and a supporting data package built for the next phase of development. |  |
Ready to explore what’s possible for your program?
Partner with ElevateBio®
Wherever you are in your advanced therapy product lifecycle, we can strengthen and accelerate your development journey with our enabling technologies and unmatched process development and manufacturing capabilities.
// Work with us
