Manufacturing & Discovery Services
We are more than a traditional CDMO. We help biopharmaceutical companies design, develop, and manufacture therapies faster and more efficiently.



// Manufacturing &
Development Services
Scaling New Heights, From Discovery to Commercialization
We provide biopharmaceutical partners with turnkey, scalable access to critical capabilities across the full product lifecycle.
Our capabilities transform innovative ideas into realities:
- Gene Editing Design & Optimization
- Clinical and Commercial Manufacturing
- Process Development
- Analytical Development
- Next-Generation Sequencing
- Regulatory and Quality Expertise
Elevated Insights
From gene editing to biomanufacturing, learn more about the cell and gene therapy industry from our team of industry-leading experts.
Modalities Powered by ElevateBio
Cell
Therapies
Viral
Vectors
mRNA Therapeutics & Vaccines
We support the design, development and manufacturing of advanced therapies across cell therapies, viral vectors, and mRNA therapeutics and vaccines. We enable partners across the full product lifecycle, from preclinical development through pivotal clinical trials and commercialization.
Learn more about
Gene Editing Design & Optimization
Move from a gene editing target to an optimized therapeutic candidate with the data to support transition to the clinic.
Process Development
Develop a scalable, robust manufacturing process built for the clinic from day one — so what works in development works at commercial scale.
cGMP Manufacturing and Automation
Manufacture your program from preclinical development through commercial scale without disruption.
Next-Generation Sequencing
Get the sequencing data your program needs to make informed development decisions, faster.
Analytical Development
Characterize your product with confidence at every stage — with custom assays and testing strategies designed to support regulatory filings.
Regulatory CMC
Navigate CMC regulatory strategy from IND through BLA with personalized support from an experienced team.
Quality
Meet the standards regulators and leaders expect — with a quality framework built specifically for genetic medicines.
Generative AI
We accelerate CRISPR discovery, combining our protein engineering expertise with a vast protein library to optimize editing systems for thousands of previously untreatable genetic disorders.
Why ElevateBio
The Difference Speaks for Itself
Partner Testimonials
We are demonstrating our focus on improving the patient experience, as well as their caregivers. Together with our partners, like ElevateBio, we look forward to continuing to innovate.
Collaboration with ElevateBio lets us expedite analytical and process development as well as outsource all manufacturing, and we can leverage their cell biology expertise throughout the process. Just ten months after launch, we had a baseline manufacturing process that took about one year off the time to IND.
The speed and quality of this collaboration exceed our expectations. ElevateBio didn’t just manufacture our therapy – they helped us establish a blueprint for base editing manufacturing.
- 98%
Batch Success Rate in 2025 - 30+
Preclinical and Clinical Programs Supported - 10+
Advanced Therapy Modalities Enabled - ICMC™
Commercial Readiness Certified - 5
Editing Modalities Supported by AI-Enzyme Discovery - 10B+
Protein Collection That Can Be Mined for Functionality
Our Technologies
Our tech-forward approach is the foundation of everything we build. From precise gene editing tools to AI-driven design, we develop and integrate the core technologies that help partners bring the next generation of advanced therapies to life.
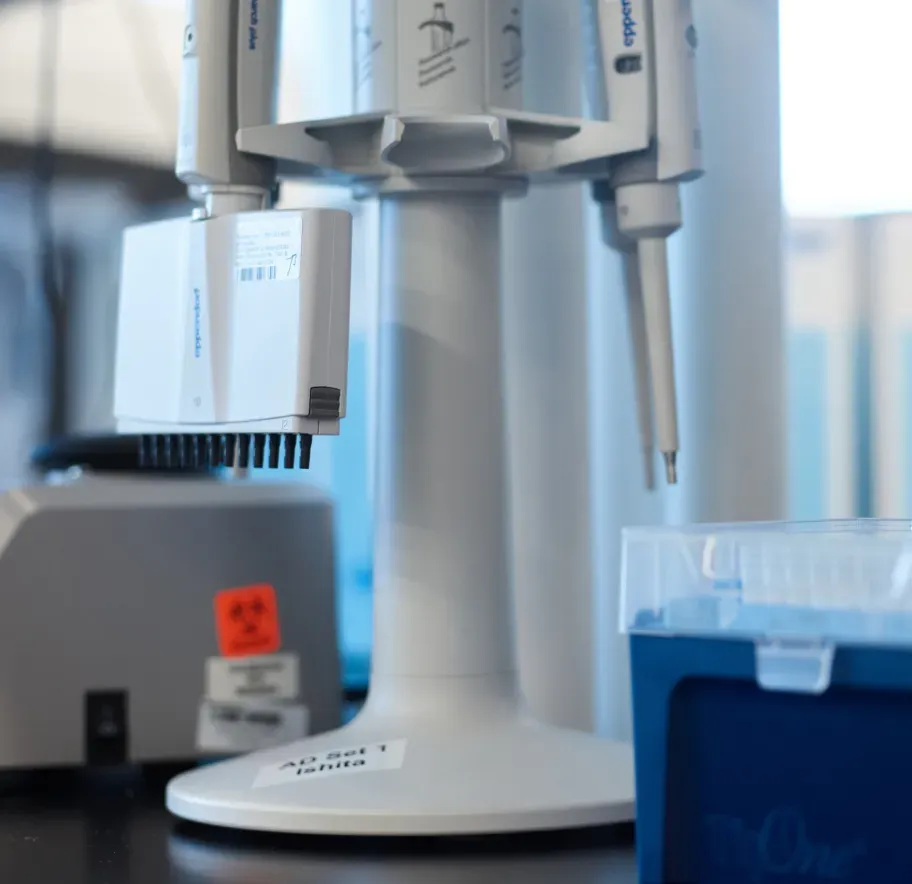

Partner with ElevateBio®
Wherever you are in your advanced therapy product lifecycle, we can strengthen and accelerate your development journey with our enabling technologies and unmatched process development and manufacturing capabilities.
// Work with us

